We provide laboratory-developed
testing approved by CLEP

The quality and performance of a laboratory test are a matter of vital concern.
CLEP approval represents accuracy that every physician should demand of laboratory tests, without compromise.
High-quality testing leads to high-quality patient outcomes.
 Newer diagnostic testing developed with advanced science and technology provides greater accuracy, facilitating better patient care. Yet with tens of thousands of laboratory-developed tests (LDTs) available, the risk of inadequate test performance can contribute to improper patient diagnosis and treatment, directly affecting patient outcomes.
Newer diagnostic testing developed with advanced science and technology provides greater accuracy, facilitating better patient care. Yet with tens of thousands of laboratory-developed tests (LDTs) available, the risk of inadequate test performance can contribute to improper patient diagnosis and treatment, directly affecting patient outcomes.
With limited oversight prior to availability for use, LDTs are excluded from regulatory requirements and have been for over 40 years, reflecting the risks these tests can pose. The FDA has identified problems with several LDTs that put patients at risk of receiving delayed or unnecessary treatment or not receiving any treatment.
Testing is the first step toward healing, and when it comes to patient health and safety, the quality of diagnostic testing is not a compromise to make.
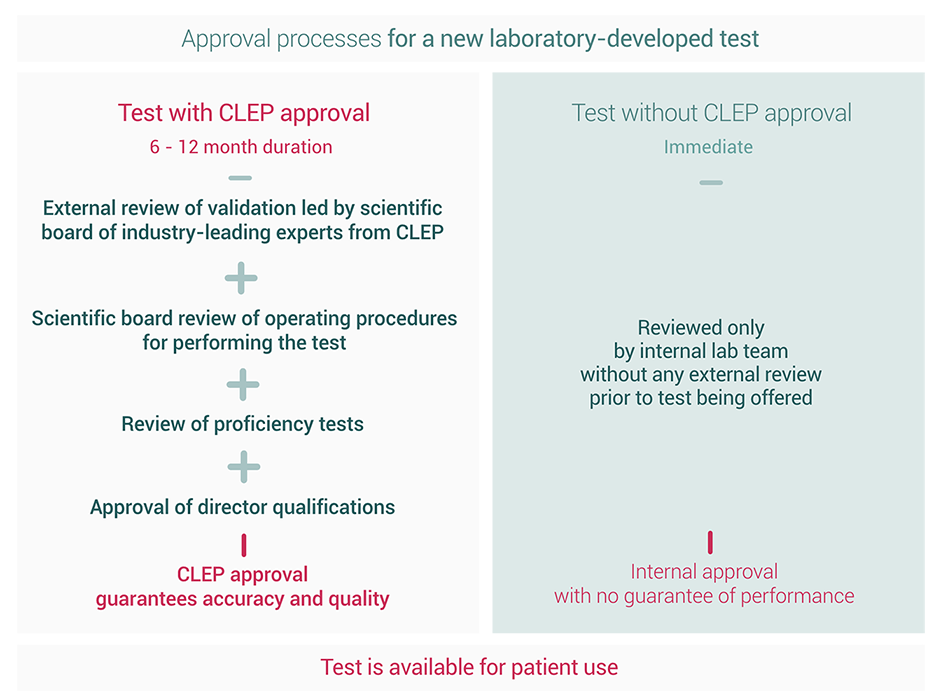
 As a specialized medical laboratory, we know clinicians require reliable and accurate testing to confidently determine the best course of treatment. That is why Acutis Diagnostics is part of less than 1% of labs in the United States providing laboratory-developed testing approved by CLEP, a third-party reviewer for the FDA, and a division of the New York Department of Health (NYSDOH). CLEP – the Clinical Laboratory Evaluation Program – oversees laboratory-developed tests to ensure accuracy and reliability.
As a specialized medical laboratory, we know clinicians require reliable and accurate testing to confidently determine the best course of treatment. That is why Acutis Diagnostics is part of less than 1% of labs in the United States providing laboratory-developed testing approved by CLEP, a third-party reviewer for the FDA, and a division of the New York Department of Health (NYSDOH). CLEP – the Clinical Laboratory Evaluation Program – oversees laboratory-developed tests to ensure accuracy and reliability.
For an LDT to meet CLEP standards, it is put through significantly more rigorous validation measures and reviews than other LDTs over an extended period of time. Most significantly, all of this is executed before it’s approved for patient use, ensuring the highest quality LDT testing in the country.
We’ve seen first-hand that our partner physicians see better patient outcomes
when using the most advanced science and testing that Acutis offers.
Our CLEP-approved tests meet stringent review standards and demonstrate clinical validity before they are authorized for patient use, enabling our practitioners to diagnose and treat patients with greater certainty and ease.